%20lewis%20structure.jpg)
It is important to note that there is no asymmetry in the C2H2 Molecular Geometry, since all of the atoms are in the same plane. Bonding pairs of electrons are those that engage in bond formation, while lone pairs or non-bonding pairs of electrons are those that do not. The Lewis Structure of any molecule helps in understanding the atomic arrangement, valence electrons, and bond formation in the molecule. The Lewis structure of C2H2 aids in the comprehension of the molecule’s shape. Furthermore, carbon bonds to carbon, giving acetylene a linear structure and a 180° bond angle. C2H2 is a tetratomic molecule made up of two distinct atoms that link in equal amounts. The Valence Shell Electron Pair Repulsion (VSEPR) theory states that the valence electrons surrounding an atom in a pair reject each other until they achieve an arrangement where this repulsion is minimised the greatest, which can be used to study the C2H2 molecular geometry. 
The angles between bonds formed by an atom are only weakly dependent on the rest of the molecule therefore, they can be thought of as roughly local and thus transferrable features. This is because the reactivity, polarity, colour, biological activity, and magnetism of a substance are all influenced by molecular geometry. Studying a molecule’s molecular geometry is a crucial step in chemistry for analysing a molecule’s behavioural features. It contains the molecule’s overall form, bond lengths, bond angles, torsional angles, and any other geometrical characteristics that govern each atom’s position. The three-dimensional arrangement of the atoms that make up a molecule is known as molecular geometry. Lastly, it has a melting point of around -80.8 degrees Celsius (or 192.3 Kelvin).C2H2 is very flammable and explosive because it is reactive and unstable. 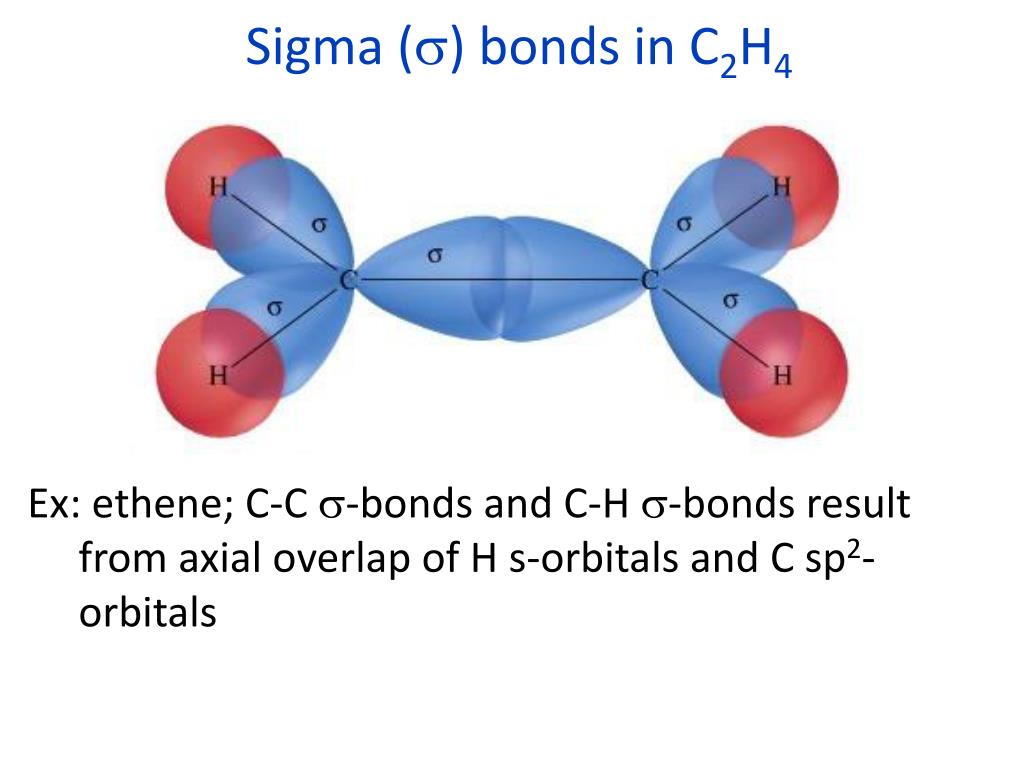
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
